Magnesium helps bacteria to survive antibiotics
Magnesium helps bacteria to survive antibiotics
The results provide a new perspective for the study of new antibiotics or even for increasing the effectiveness of currently used antibiotics.

Antibiotics are our main strategy to fight bacterial infections. Sometimes, however, when we apply an antibiotic to a population of bacteria, not all of them are eliminated. Even when all cells in the population are genetically identical, have not developed a resistance to antibiotics and are found in identical conditions, some bacteria survive and others do not. Why does this occur? Now, scientists at Pompeu Fabra University in Barcelona and the University of California San Diego have discovered that this situation is caused by magnesium, which promotes the survival of bacteria against certain types of antibiotic.
In this work, published in Cell, the researchers studied antibiotics that fight bacteria by blocking their production of proteins. That is, they attack ribosomes, the molecular machinery responsible for protein production within cells, and therefore one of the cornerstones for bacterial growth. Ribosomes maintain their complex structure due to the presence of ions, electrically charged molecules.
Els ions que tenen el paper més important en l’estabilitat del ribosoma són els de magnesi, ja que s’hi uneixen i els aporten cohesió.
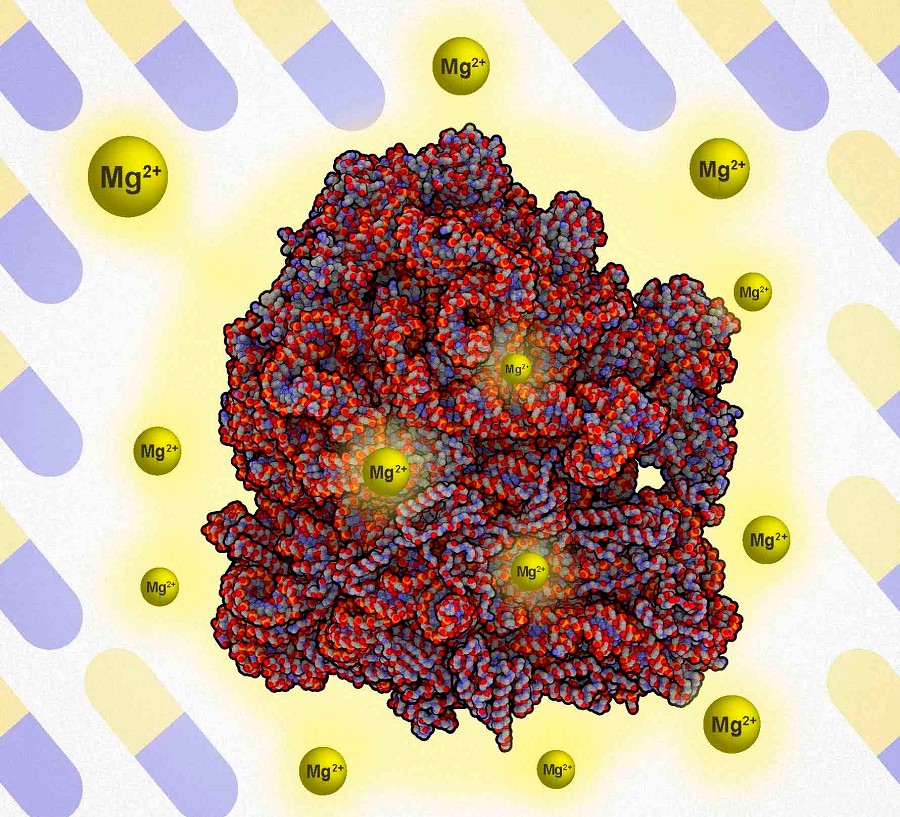 The scientists found that the ions that play the most important role in the stability of the ribosome are magnesium ions, as they bind to the ribosomes and provide them with cohesion. “So, when we attack the ribosomes with antibiotics, if the former have sufficient magnesium, they are more stable and therefore more resilient”, explains Jordi Garcia-Ojalvo, full professor of Systems Biology with the Department of Experimental and Health Sciences (DCEXS) at UPF. “However, when the bacteria have little magnesium, their ribosomes are less robust and when they are attacked they destabilize, so that the cell cannot produce proteins properly and dies”, he adds.
The scientists found that the ions that play the most important role in the stability of the ribosome are magnesium ions, as they bind to the ribosomes and provide them with cohesion. “So, when we attack the ribosomes with antibiotics, if the former have sufficient magnesium, they are more stable and therefore more resilient”, explains Jordi Garcia-Ojalvo, full professor of Systems Biology with the Department of Experimental and Health Sciences (DCEXS) at UPF. “However, when the bacteria have little magnesium, their ribosomes are less robust and when they are attacked they destabilize, so that the cell cannot produce proteins properly and dies”, he adds.
Letícia Galera-Laporta, first author of the study, explains that they “combined the study of a mathematical model and experiments, including various chemical and structural disturbances to the ribosomes, to show that the main factor that determines the survival of the bacterium Bacillus subtilis to antibiotics is magnesium flux regulation”.
It is technically difficult to measure the concentration of magnesium in individual live cells, but the scientists were able to obtain this data by measuring the membrane potential of millions of cells using fluorescence microscopy. Membrane potential is present in all cells, such as the neurons in the brain, and is associated to the difference of ions inside and outside the cell.
This property allowed the scientists to detect two types of cells within the same population: some cells present sudden changes in membrane potential, since they do not properly regulate the ion flux through their membrane, and end up dying. On the other hand, cells that manage to modulate the magnesium ion flux and keep their membrane potential stable under the stress of the antibiotic, are able to survive the attack.
“Our results provide a new perspective for the study of new antibiotics or even for increasing the effectiveness of currently used antibiotics. One possible line of future research, for example, could be to use antibiotics together with a supplement that blocks the transport of magnesium to bacteria”, concludes Letícia Galera-Laporta.
Reference article:
Dong-yeon D. Lee, Leticia Galera-Laporta, Maja Bialecka-Fornal, Eun Chae Moon, Zhouxin Shen, Steven P. Briggs, Jordi Garcia-Ojalvo and Gürol M. Süel. Magnesium flux modulates ribosomes to increase bacterial survival. Cell, March 2019. DOI: 10.1016/j.cell.2019.01.042.
