Virus-host interactions
Department of Medicine and Life Sciences
Edifici PRBB (campus del Mar)
Doctor Aiguader, 88
08003 Barcelona
+34 933160862
Principal investigator: Andreas Meyerhans
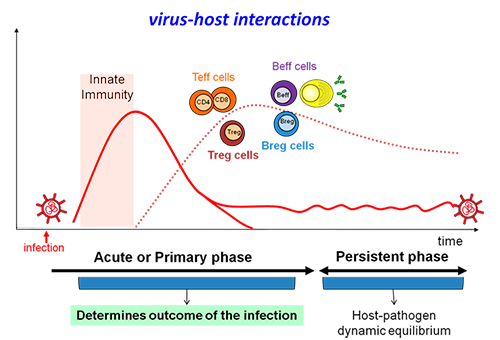
The dynamic interactions between a virus and its host are critical for the establishment and maintenance of persistent infections. However, how the shift from acute to a chronic infection is regulated and how the immune response influences virus control, is still poorly understood. In the previous years we have established in the laboratory (1) a Lymphocytic Choriomeningitis Virus (LCMV)-infection mouse model system that enables to direct the infection fate (acute or persistent infection) on the basis of the initial virus infection dose or virus strain, and (2) experimental tools to study the functionality of antigen-specific, effector T lymphocytes including their exhausted counterparts and regulatory T lymphocytes in human infections. With these in hand, we will address 2 fundamental issues in the coming years. First, we will use the LCMV-infection mouse model system and analyse spleen-specific transcriptomes of infected mice that exhibit different infection outcomes with the objective to identify key features in the fate decisions between acute and persistent infections. Second, we will quantify lymphocyte down-regulation and exhaustion in persistently Human Immunodeficiency Virus (HIV)-infected humans with the objective to better characterize the relationship of lymphocyte responsiveness and virus control, thus providing anti-exhaustion therapy indicators. We expect that the obtained results will help to (1) identify mechanisms that determine infection outcomes and (2) gain detailed insights into infection control mechanisms and new therapeutic modalities.
