Research
CURRENT PROJECTS

Otic Placode Morphogenesis
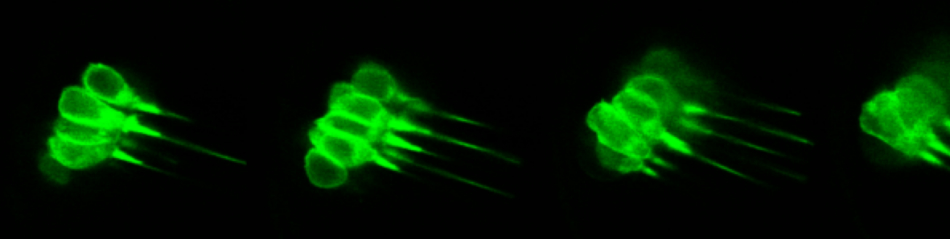
The inner ear is one of the most complex three-dimensinal organs of our head, however it is still little understood how cells organize during development to generate this complex organ. We are studying several morphogenetic events to understand the interaction between cell polarity, cell remodeling, migration and cytoskeleton rearrangements with mechanical properties and signalling cues.
Otic precursors are initially not organized, cells have arounded shape and progressively orient and elongate to form a ordered epithelium. The sequence of events involving the establishment of apical polarity, microtubule orientation, actomyosion activation, ECM and rosette formation is unknown. We aim to understand the cellular remodelling events during placode formation and the biomechanics driving the formation of an ellipsoidal placode.

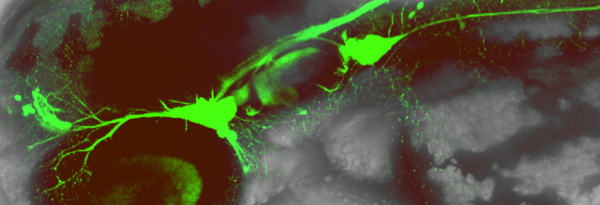
Hair Cell innervation and Mechanosensory circuit formation
How SAG axons are directed towards inner hair cells, how the SAG shape is established? Furthermore, how statoacoustic axons enter the hindbrain? We are addressing all these questions to understand how the mechanosensory circuit is established

Transcription factors and chromatin remodelling in otic neuron identity
The identification of TF driving neuronal differentiation and subtype sensory identities is crucial for regenerative medicine therapies and for the understanding the basis of sensory diseases. Compared to the central nervous system and the development of hair cells, very little is known of how the transition from inner ear neuronal precursors to differentiated neurons is achieved at the molecular level. What makes an otic sensory neuron different from a nociceptor or chemosensory neuron?
We aim at identifying which TFs are key in otic sensory neuronal identity and maintaning their terminal features, together with the analysis of the chromatin remodelling events regulating the difference between distict sensory neuronal subtypes.
Inner ear organoids for studying mutations causing hearing loss
Several mutations either in the coding regions or regulatory elements have been linked to hearing loss, vertigo or neuropathies. In some instances, the sensory disease is not due to a malfunctioning protein but to a reduction/increased of expression or epigenetic modifications of the affected loci. Those mutations might be rescued by modification of the expression levels using the crispr system. We are testing the use of the cripr system to revert regulatory mutations using human inner ear organoids.