Molecular Physiology
Miguel Angel Valverde

Group website
Research Outline
Our research focuses on ion channels, transporters and signaling in the context of cellular responses to oxidative, mechanical and osmotic stresses. Our work aims to translate our understanding of cellular physiology into a better understanding of the mechanisms of human disease.
Research Lines
MECHANO/OSMOSENSITIVE ION CHANNELS IN SHORT- AND LONG-TERM CELLULAR FUNCTIONS (MECHANICAL)
Mechanical forces influence cell behavior, from cell shape and movement to cell division and gene transcription through mechanotransduction and mechanoresponsive pathways triggered by cells. Understanding how cells dynamically integrate mechanical signals from the microenvironment is a challenging question in cell biology. So far, force and geometry sensing focuses on cell interaction with extracellular matrix via integrin-based complexes and the closely associated actomyosin cytoskeleton. However, cell behavior under defined 3D environments may also show integrin- and actomyosin-independent transducing mechanisms. The novelty of our proposal is to place Mechano/osmosensitive (MOS) Ion Channels central to both integrin-dependent and -independent processes. We will address several basic questions related to the mechanisms of activation/regulation of MOS channels as well as their biological/medical relevance in
long-range (term) cellular responses such as cancer proliferation, invasion and metastasis as well as in RNA metabolism and viral infection.
Team during 2019-20
PhD students: Julia Carrillo Garcia, Victor Herrera Fernandez
Postdocs: Selma Serra Pascual, Fanny Rubio Moscardo, Pau Doñate Macian
Technicians: Cristina Plata Fernández
Selected publications 2019-20
- Cross-talk between the calcium channel TRPV4 and reactive oxygen species interlocks adhesive and degradative functions of invadosomes. Vellino S, Oddou C, Rivier P, Boyault C, Hiriart-Bryant E, Kraut A, Martin R, Coute Y, Knölker HJ, Valverde MA, Albigès-Rizo C, Destaing O.J Cell Biol. 2021 Feb 1;220(2):e201910079. doi: 10.1083/jcb.201910079.PMID: 33399853
- The nucleus measures shape changes for cellular proprioception to control dynamic cell behavior. Venturini V, Pezzano F, Català Castro F, Häkkinen HM, Jiménez-Delgado S, Colomer-Rosell M, Marro M, Tolosa-Ramon Q, Paz-López S, Valverde MA, Weghuber J, Loza-Alvarez P, Krieg M, Wieser S, Ruprecht V. Science. 2020 Oct 16;370(6514):eaba2644. doi: 10.1126/science.aba2644.PMID: 33060331
- Structural determinants of TRPV4 inhibition and identification of new antagonists with antiviral activity. Doñate-Macian P, Duarte Y, Rubio-Moscardo F, Pérez-Vilaró G, Canan J, Díez J, González-Nilo F, Valverde MA. Br J Pharmacol. 2020 Sep 21. doi: 10.1111/bph.15267. Online ahead of print.PMID: 32959389
- Cell sensing and decision-making in confinement: The role of TRPM7 in a tug of war between hydraulic pressure and cross-sectional area. Zhao R, Afthinos A, Zhu T, Mistriotis P, Li Y, Serra SA, Zhang Y, Yankaskas CL, He S, Valverde MA, Sun SX, Konstantopoulos K.Sci Adv. 2019 Jul 24;5(7):eaaw7243. doi: 10.1126/sciadv.aaw7243. eCollection 2019 Jul.PMID: 31355337
- Sodium channel TRPM4 and sodium/calcium exchangers (NCX) cooperate in the control of Ca2+-induced mucin secretion from goblet cells. Cantero-Recasens G, Butnaru CM, Brouwers N, Mitrovic S, Valverde MA, Malhotra V.J Biol Chem. 2019 Jan 18;294(3):816-826. doi: 10.1074/jbc.RA117.000848. Epub 2018 Nov 27.PMID: 30482841
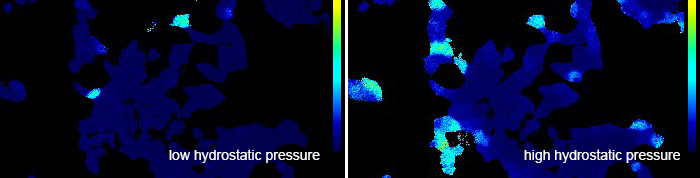
Cells overexpressing the TRPM7 cation channel increase the intracellular calcium concentration when exposed to high hydrostatic pressure.
