Optimisation can help increasing swab test capacity
Optimisation can help increasing swab test capacity
Links available to the dashboard, the technical report, and the source code and data.
Large-scale testing of the population is a key tool to limit the spread of the disease. Governments must set up massive test programmes and, to contain new virus clusters, they must test often and test quickly.
Standard PCR test kits work by taking a swab with a sample of saliva from a person, and looking for viral RNA on the swab. For the virus's RNA to be identifiable, it must first be transformed into DNA (i.e., it must go from single to double helix) and then replicated, so that it reaches detectable quantities. Laboratories need reagents to carry out the steps of this procedure.
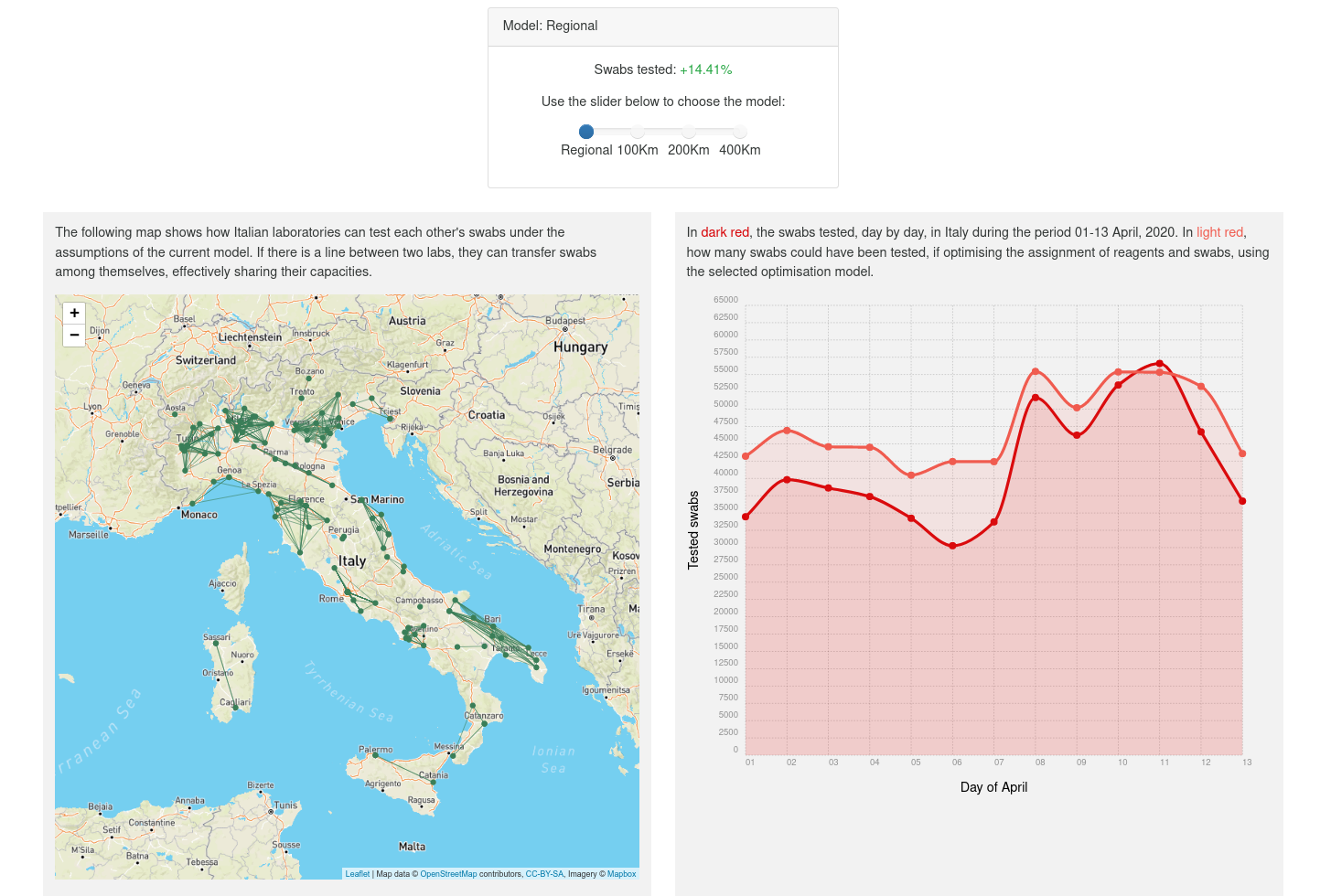
During the COVID-19 pandemic, reagents availability has been a major bottleneck to increase test capacity. We show that we can optimise two decisions which lead to a noticeable increase in capacity:
- How to allocate reagents to the laboratories, and
- whether to send swabs collected in one region to labs in other regions.
We use the Italian healthcare system as a case study. The healthcare system in Italy is organised on a regional basis and cross-regional collaboration isn't as developed as it could. We argue that this results in missed optimisation opportunities, and we provide some data to support this hypothesis.
